


Next: Wave-Particle Duality
Up: The Motivation for Quantum
Previous: The Photoelectric Effect
Rutherford proposed that electrons orbit about the nucleus of an
atom. One problem with this model is that, classically, orbiting
electrons experience a centripetal acceleration, and accelerating charges
lose energy by radiating; a stable electronic orbit is classically
forbidden. Bohr nevertheless
assumed stable electronic orbits with the electronic angular momentum
quantized as
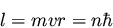 |
(2) |
Quantization of angular momentum means that the radius of the orbit
and the energy will be quantized as well.
Bohr assumed that the discrete lines seen in the spectrum
of the hydrogen atom were due to transitions of an electron from one
allowed orbit/energy to another. He further assumed that
the energy for a transition is acquired or released in the form of a
photon as proposed by Einstein, so that
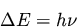 |
(3) |
This is known as the Bohr frequency condition. This condition,
along with Bohr's expression for the allowed energy levels, gives a
good match to the observed hydrogen atom spectrum. However, it works
only for atoms with one electron.



Next: Wave-Particle Duality
Up: The Motivation for Quantum
Previous: The Photoelectric Effect